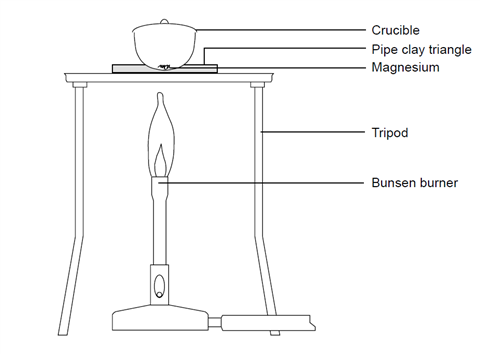
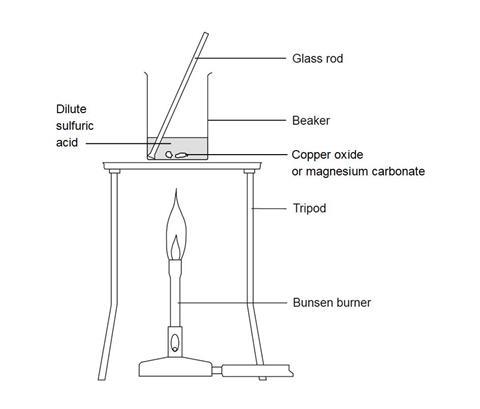
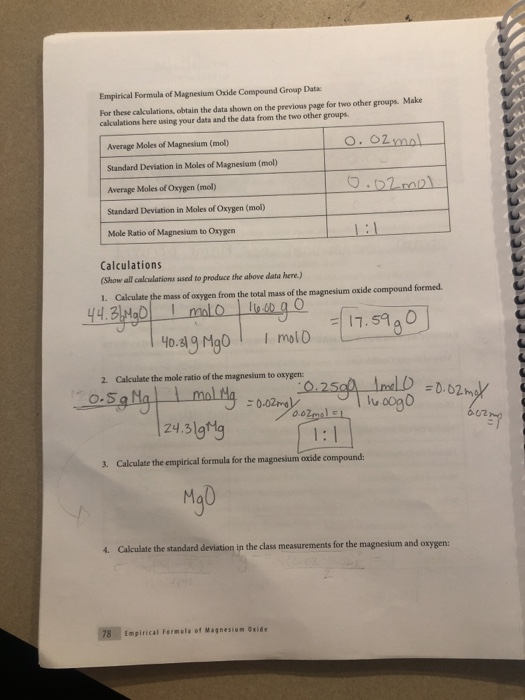
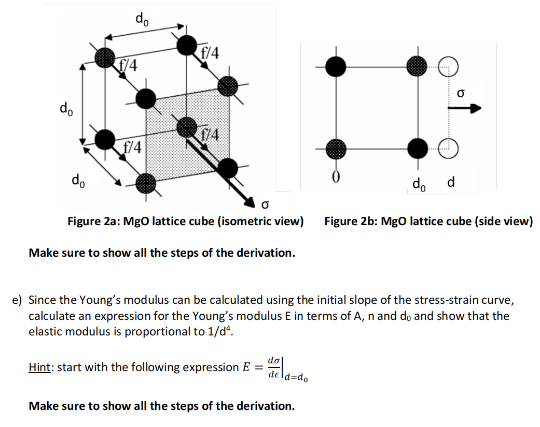
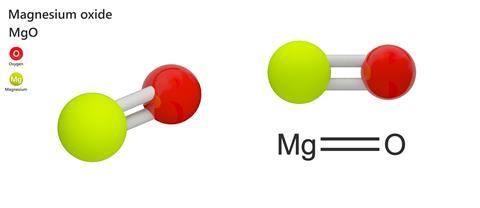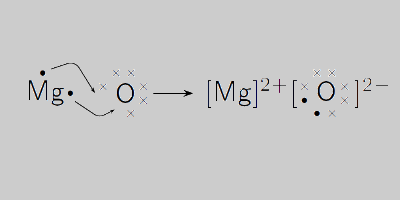Magnesium and carbon dioxide – Student sheet Burning magnesium in carbon dioxide – what will happen? To do
A 3.250g sample of magnesium is burned in a container of 12.500g oxygen. What mass of oxygen gas remains unreacted after the magnesium has been completely consumed to form magnesium oxide as